WARNING: this project has been retired. Please migrate to geem-lab/overreact.
pyrrole¶
A Python package for solving chemical problems with computational modeling.
Usage example¶
As a usage example, let’s calculate the energy barrier involved in nitrogen inversion in ammonia.
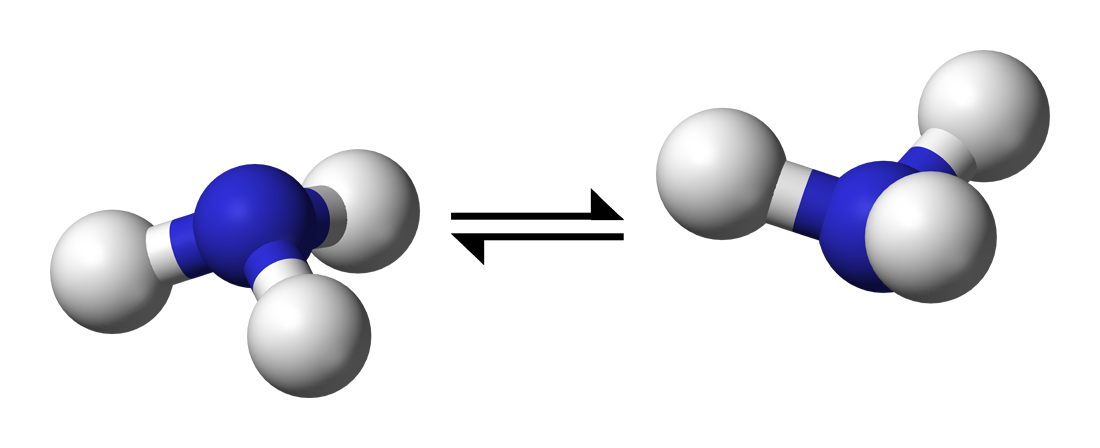
When ammonia turns “inside out”, it passes through a planar transition state (image in public domain).
We do this in three simple steps (only eight lines of code):
- Get the data
We first obtain the raw data, which will later be fed to our chemical model. Below we read computational chemistry logfiles of both ground and transition states [1].
>>> from pyrrole.atoms import read_cclib, create_data
>>> gs = read_cclib("data/ammonia/ammonia.out", name="NH3(g)")
>>> ts = read_cclib("data/ammonia/invers.out", name="NH3(g)#")
>>> data = create_data(gs, ts)
Pyrrole uses cclib for reading logfiles, which is compatible with all major computational chemistry packages. You could also want to read tabular data from a file (or even from the web) using pandas.
- Specify the model
We now describe our model. This is accomplished through chemical equations:
>>> from pyrrole import ChemicalEquation
>>> equation = ChemicalEquation("NH3(g) -> NH3(g)#", data)
While model above consists of a single ChemicalEquation, you could create complex models with multiple chemical equations with ChemicalSystem objects.
You might want to store your complex models in text files too.
- Obtain the results
Simply let pyrrole calculate the energy barrier:
>>> results = equation.to_series()
>>> results["freeenergy"] * 2625.4996382852164 # Hartree to kJ/mol
19.30952589472923
(As a side note, the reference value is 21.162 kJ/mol [2].)
Interested? Have another example.
| [1] | Optimizations and frequency calculations of both ammonia and the planar transition state were performed at PBEh-3c using the ORCA electronic structure package (version 4.0.1.2). Logfiles can be found in the project’s repository. |
| [2] | Chem. Phys. Lett., 2003, 370 (3), pp 360-365 DOI: 10.1016/S0009-2614(03)00107-6. |